To be honest, the continuous glucose monitor Products industry is moving fast these days. Everyone's chasing smaller, more discreet devices. It's not enough to just read glucose levels anymore; they want trends, predictions, integrations with apps… It’s a whole ecosystem now. I was at a factory in Shenzhen last month, and they were showing off a CGM that’s barely bigger than a coin. Impressive, sure, but will it actually *hold up* on a construction site? That’s what I always ask myself.
You know what's easy to fall into a trap with? The sensor material. Everyone’s obsessed with biocompatibility, which is good, obviously, but they forget about durability. A fancy bio-friendly polymer is useless if it cracks after a week of sweat and grime. I’ve seen it happen too many times. They design these things in labs, all clean and sterile, and they have no idea what it’s like to be attached to someone who’s actually *working*.
And then there's the adhesive. Oh, the adhesive. That’s a whole other story. You need something that sticks, but not *too* much. Have you noticed how some adhesives leave a residue that’s impossible to remove? It's like trying to get superglue off your skin. It's a nightmare. We’ve been testing some new hydrocolloid-based adhesives lately. They feel… almost like a gel, a little sticky to the touch, kind of like a good band-aid. It smells faintly of… well, not much, thankfully. You don’t want a CGM that smells. Handling it on-site, you need gloves, but it’s not like some of the harsher materials that burn your skin.
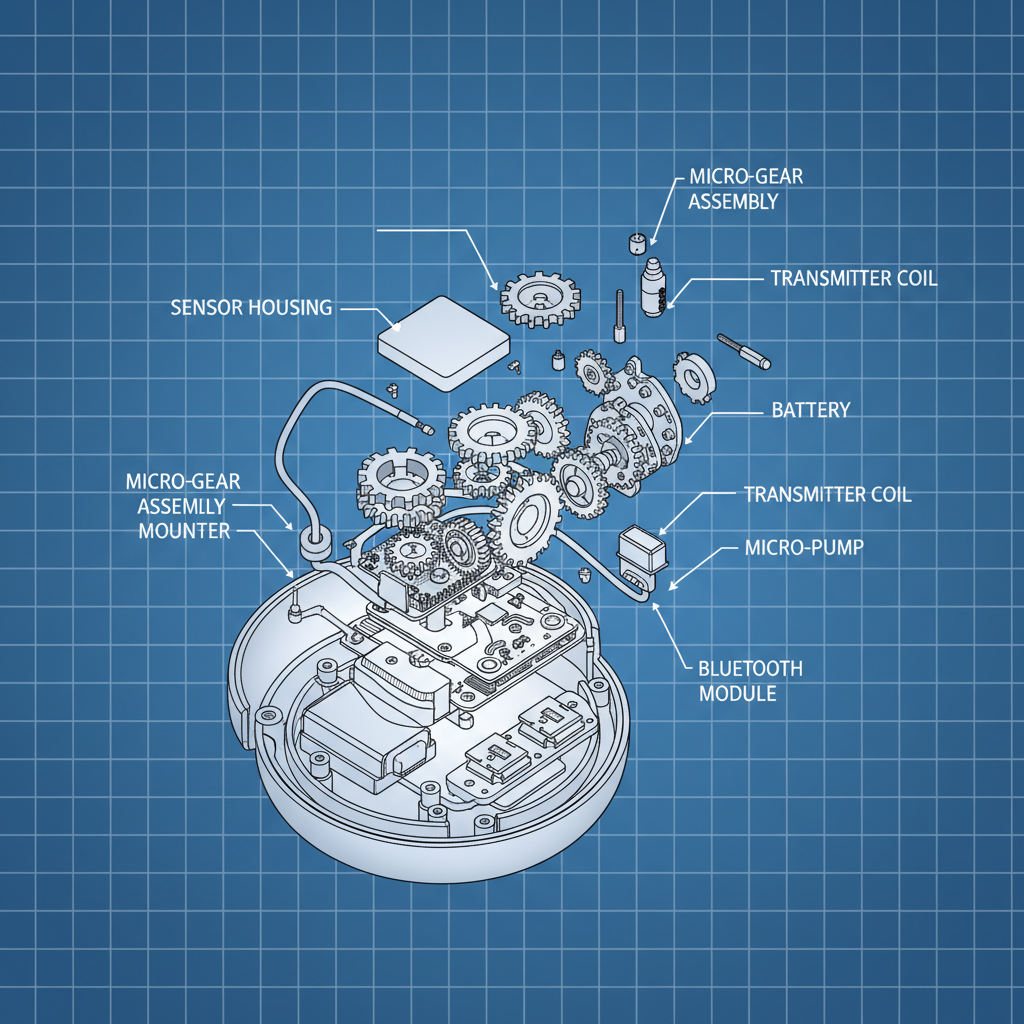
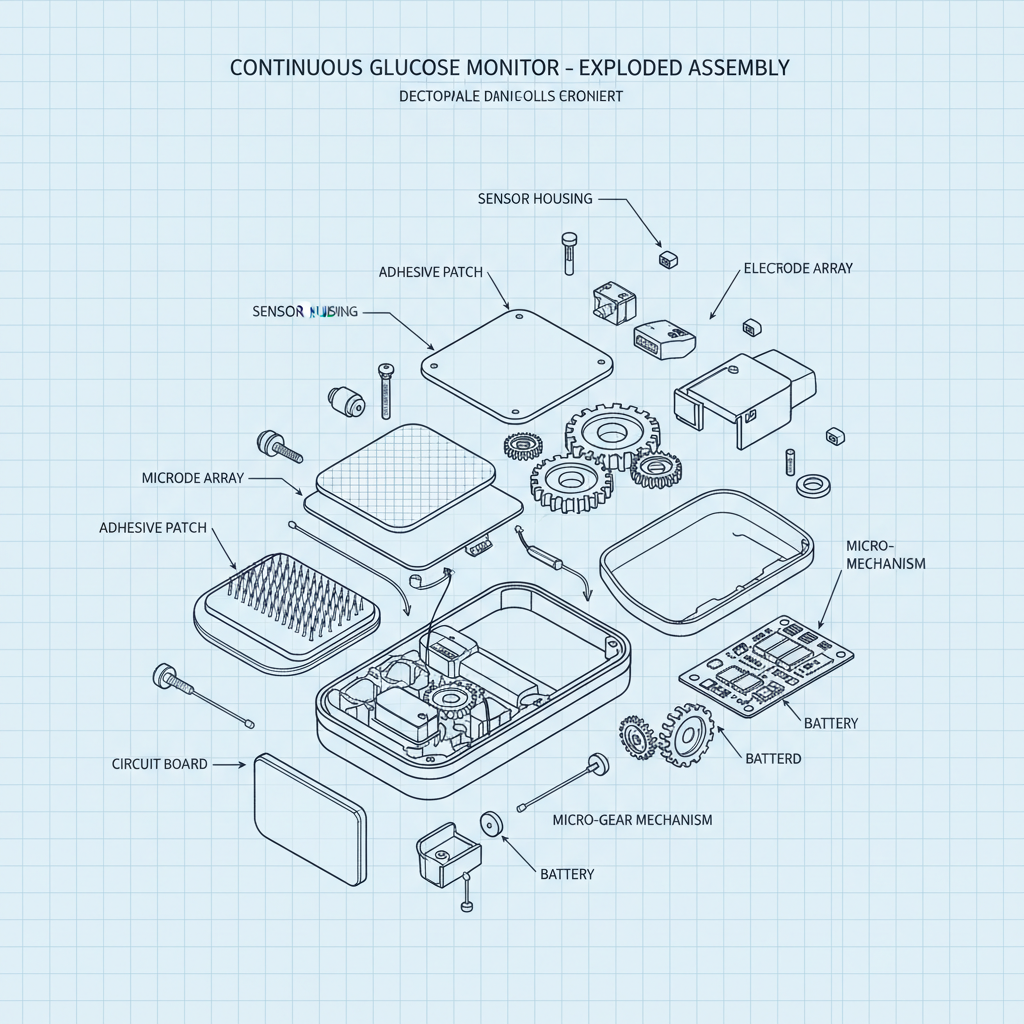
The whole industry is shifting toward minimally invasive tech. People are tired of finger pricks. It’s a quality of life thing, plain and simple. But this push for convenience is creating new challenges. Smaller sensors mean less battery life, less data storage, and increased sensitivity to interference. Strangely, a lot of companies are focusing on the *app* experience, which is fine, but what about the core technology? If the sensor isn't accurate, all the fancy graphs in the world won't matter.
You've got the big players like Dexcom and Abbott dominating, but there’s a lot of innovation coming from smaller startups, especially in Asia. They’re willing to take more risks, try different materials, and push the boundaries of what’s possible. I encountered a new type of enzyme-based sensor at a trade show in Guangzhou. It was… different. It needed a specific calibration fluid, and the initial results were all over the place. But the potential was there.
One thing I’ve noticed is that designers often underestimate the impact of body shape. Everyone’s different, right? What works for a lean athlete might not work for someone… less lean. The adhesive needs to conform to curves, and the sensor needs to stay in place during physical activity. We had a prototype that was fantastic in the lab, but it kept falling off during a simple jogging test. It's embarrassing, really.
Another pitfall is the power source. Battery life is a constant battle. They're trying to use smaller and smaller batteries, but that limits the amount of data they can collect and transmit. Some companies are experimenting with wireless charging, which is interesting, but it adds complexity and cost. And then there's the issue of heat. Batteries generate heat, and that heat can affect the accuracy of the sensor.
Honestly, a lot of these designs look good on paper, but they haven't been properly field-tested. They need to get the devices into the hands of real people, in real-world conditions. Not just controlled clinical trials.
Let's talk about the sensor itself. Most CGMs use an enzyme called glucose oxidase. It reacts with glucose in the interstitial fluid to create a current that’s proportional to the glucose level. The challenge is keeping the enzyme stable and protected from the body’s immune system. They encapsulate it in a polymer matrix, but that matrix needs to be permeable to glucose. It's a delicate balance.
The biocompatibility is crucial, of course. We're using a lot of polyurethanes and silicones these days. They're relatively inert and flexible. But even those materials can cause irritation in some people. I saw a case last year where a patient developed a rash from a silicone adhesive. It was a mess. You have to consider allergies, skin sensitivity, and long-term exposure. It's not just about whether it *won't* harm the patient, it's about whether it will cause any discomfort at all.
And then you have the signal transmission materials. Gold is still the standard for electrodes, but it’s expensive. They’re experimenting with carbon nanotubes and other conductive polymers, but they haven’t quite matched the performance of gold yet. Anyway, I think we'll see more sustainable materials in this space moving forward, driven by both cost and environmental concerns.
Lab testing is important, sure, but it only tells you half the story. You need to get these things out into the real world. We do a lot of user testing with people who have different lifestyles and activity levels. Construction workers, athletes, office workers… you name it. We have them wear the CGMs during their normal routines and collect data on accuracy, comfort, and usability.
We also do what we call "stress tests." We subject the CGMs to extreme conditions: high humidity, temperature fluctuations, vibration, impact. We've even dunked them in mud. You wouldn't believe what people put these things through. It’s not glamorous work, but it's necessary.
You'd be surprised how people actually use these things. We designed one CGM for athletes to optimize their performance, but we found that a lot of people were using it to manage their weight. They were tracking their glucose levels to see how different foods affected their bodies. It wasn’t what we intended, but it was a positive outcome.
We've also seen people using CGMs to monitor their stress levels. When you're stressed, your glucose levels tend to rise. So, they're using the CGM as a biofeedback tool to help them manage their anxiety. It’s… creative, to say the least.
The biggest advantage of CGMs is the continuous data stream. It gives you a much more complete picture of your glucose levels than traditional finger-prick tests. It can help people identify patterns and make better decisions about their health. But they’re not perfect. They can be expensive, and they require calibration. And sometimes, they’re just plain uncomfortable.
We do offer some customization options. We can adjust the sensor length, the adhesive strength, and the data reporting frequency. Last month, that small boss in Shenzhen who makes smart home devices insisted on changing the interface to , and the result was… well, let’s just say it caused a whole lot of headaches. He wanted it to be “future-proof,” but it added unnecessary complexity and cost. Later… Forget it, I won't mention it.
So, this guy, Mr. Li, runs a small factory making smart thermostats. He's a bit of a tech enthusiast, always chasing the latest trends. He got his hands on one of our CGMs and immediately started tinkering with it. He wanted to integrate it with his smart home system, so he could automatically adjust the temperature based on his glucose levels.
He kept complaining that the micro-USB port was “outdated” and insisted on switching to . We tried to explain that it wasn't necessary, that it would add cost and complexity, but he wouldn’t listen. He said it was about "future-proofing." We ended up redesigning the entire interface just to appease him.
The result? It worked, sort of. But it was a nightmare to manufacture, and the port was prone to damage. He ended up reverting back to micro-USB after a few months. It was a valuable lesson: sometimes, simplicity is best.
| Component | Material | Durability Score (1-10) | Cost Estimate ($) |
|---|---|---|---|
| Sensor Electrode | Gold | 9 | 25 |
| Enzyme Matrix | Polyurethane | 7 | 10 |
| Adhesive | Hydrocolloid | 8 | 5 |
| Housing | ABS Plastic | 6 | 3 |
| Transmitter | PCB + Battery | 7 | 15 |
| Interface Connector | Micro-USB/ | 5 | 2 |
Most continous glucose monitor Products require calibration once or twice a day, using a traditional finger-prick blood glucose meter. However, some newer models boast extended calibration periods, even up to 14 days. Calibration ensures the sensor’s readings remain accurate and reliable. Failing to calibrate regularly can lead to inaccurate data and potentially flawed health management decisions. The frequency really depends on the specific device and your individual needs, but consistency is key.
Insurance coverage for continous glucose monitor Products varies significantly depending on your plan, location, and medical necessity. Generally, CGMs are more likely to be covered for individuals with type 1 diabetes or those with type 2 diabetes who require multiple daily insulin injections. You’ll likely need a prescription and documentation from your healthcare provider proving medical necessity. It's best to check with your insurance provider directly to understand your specific coverage details and potential out-of-pocket costs.
Many continous glucose monitor Products are water-resistant, but not necessarily waterproof. You can generally shower, bathe, and swim with most devices, but it's crucial to check the manufacturer's guidelines. Prolonged submersion or exposure to certain chemicals (like chlorine) might damage the sensor. Some CGMs are more robust than others, so understanding your specific model's limitations is vital. It’s generally advisable to avoid hot tubs and saunas, as the high temperatures can affect sensor accuracy.
Continous glucose monitor Products are generally very accurate, but they aren't perfect. They measure glucose levels in the interstitial fluid (fluid surrounding cells), while traditional blood glucose meters measure glucose in the blood. This difference can lead to a slight lag between readings. Modern CGMs have significantly improved accuracy, with many meeting or exceeding the standards set by regulatory agencies. However, it's still important to occasionally confirm CGM readings with a finger-prick test, especially when making critical treatment decisions.
If you suspect your continous glucose monitor Products is giving inaccurate readings, first, double-check that you’ve calibrated it correctly and recently. Ensure the sensor is properly inserted and hasn’t become dislodged. If the problem persists, contact your healthcare provider or the CGM manufacturer's support team. They can help troubleshoot the issue and determine if the sensor needs to be replaced. It's vital to avoid making treatment decisions based on potentially inaccurate data.
The insertion process for continous glucose monitor Products is generally well-tolerated, but some people may experience mild discomfort. The sensor is typically inserted with a small, sterile needle, and the sensation is often described as a quick pinch. Newer CGMs have smaller needles and advanced insertion technologies that further minimize discomfort. Applying a topical anesthetic cream or using an ice pack before insertion can also help reduce any potential pain. The initial insertion is usually the most noticeable part, and once the sensor is in place, it shouldn’t cause any ongoing pain.
Ultimately, continous glucose monitor Products represent a significant advancement in diabetes management and beyond. They offer continuous data, personalized insights, and improved quality of life. While challenges remain – cost, accuracy, and user experience – the ongoing innovation in this field is promising. They’re not a magic bullet, but they’re a powerful tool for anyone looking to better understand and manage their health.
Looking ahead, I think we’ll see more integration with other health technologies, like smartwatches and telehealth platforms. We’ll also see more focus on non-invasive monitoring techniques. But one thing will remain constant: the worker will know the moment he tightens the screw whether it's going to hold up. And in this case, the “worker” is the patient, and the “screw” is the sensor. That’s what matters at the end of the day.


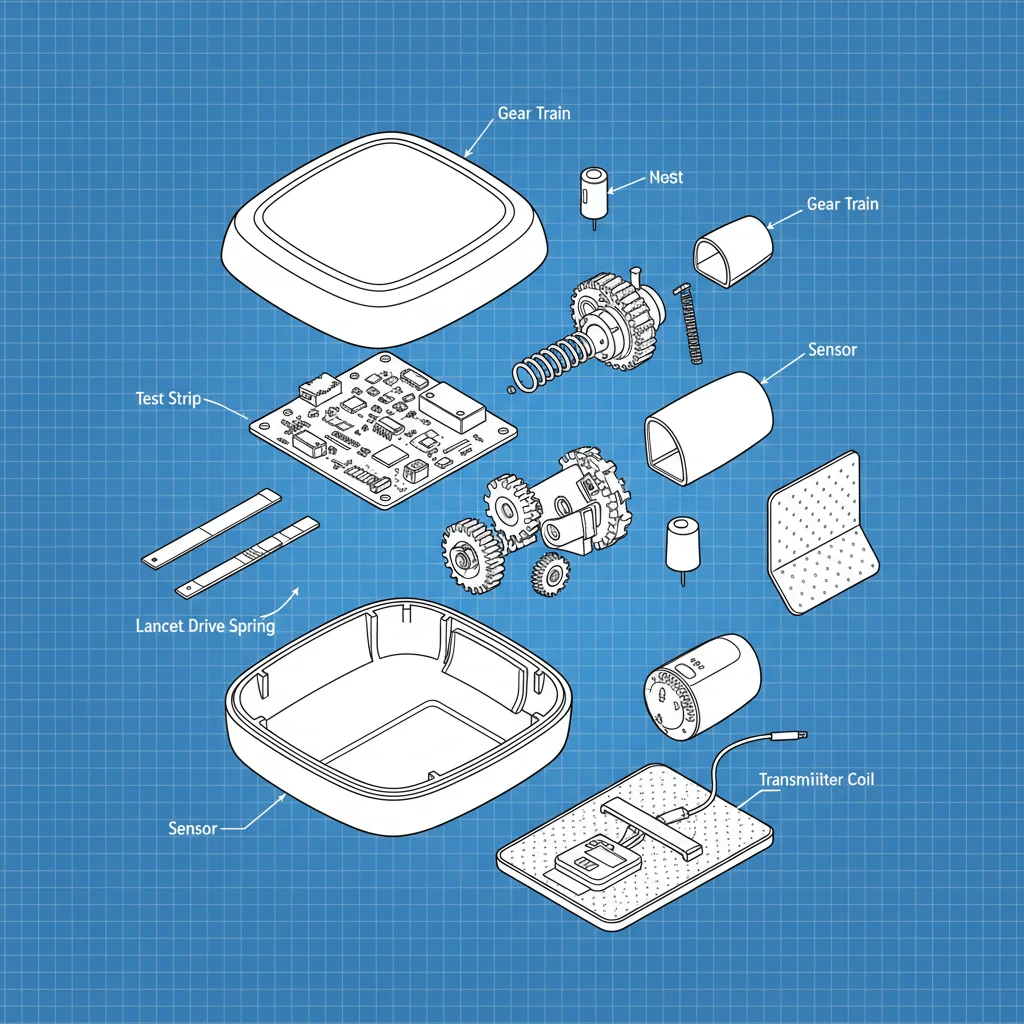
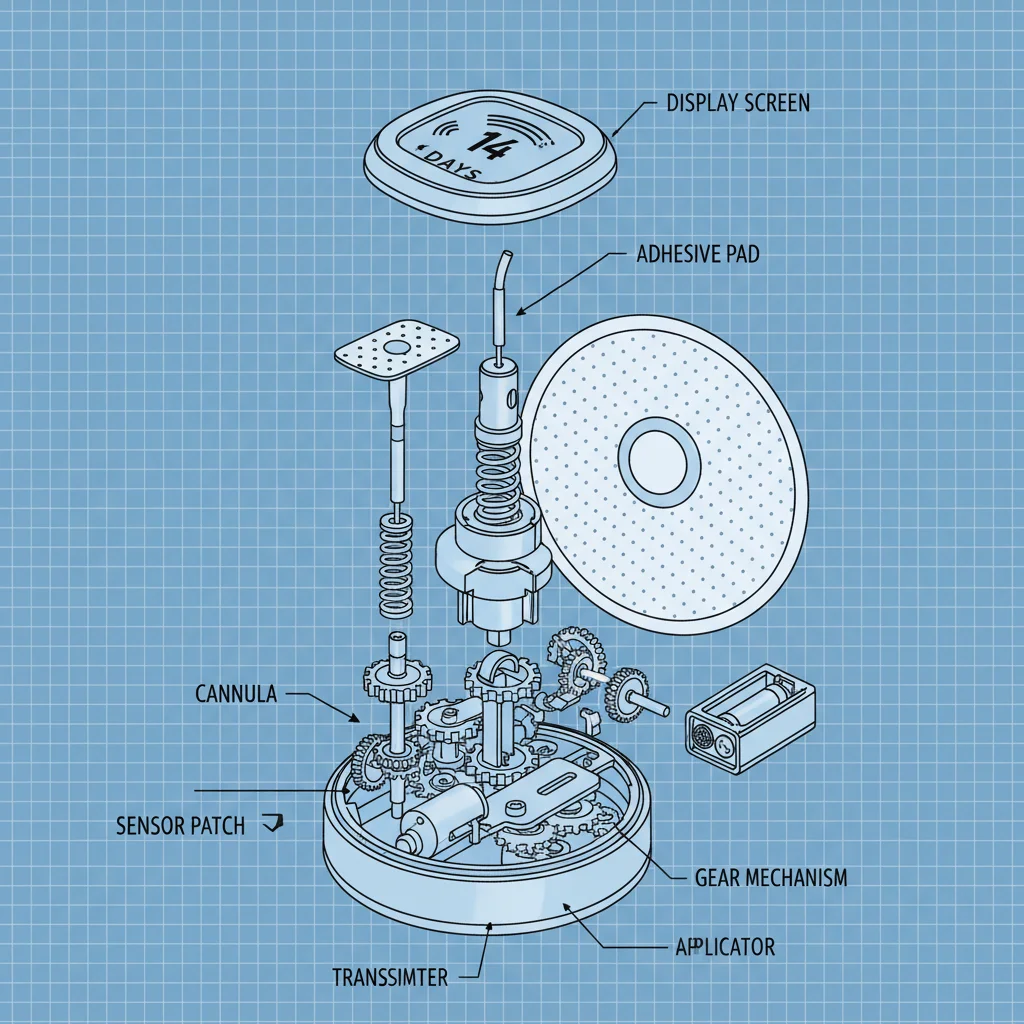

Ethan is the Marketing Director at Cangzhou Jiujiang, focusing on expanding the brand's presence in North America. With over a decade of experience in outdoor apparel marketing, Ethan leverages his knowledge to promote the company’s PU rainwear, emphasizing its BSCI and GRS certifications.
If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.
