Look, things are moving fast these days. Everyone's talking about miniaturization, right? Making everything smaller, more efficient. But honestly, sometimes I think people forget what happens when you actually *build* something. You design a tiny connector, sleek and fancy, but then try fitting it onto a vibrating machine in a dusty factory… it’s a whole different ballgame.
I’ve been seeing a lot of reliance on simulations, too. “Oh, the software says it can handle the stress.” Yeah, well, software doesn’t smell burning plastic at 3 AM. Anyway, I think we’re at a point where we need to get back to basics – robust design, real-world testing, and actually talking to the guys on the ground. It’s the only way.
And speaking of the ground, it's all about continuous glucose monitor cgm Products these days, or at least that’s what the big bosses are saying. Seems like everyone’s trying to find a better way to monitor things, track data, and improve efficiency. I mean, it makes sense, but… it’s also complicated.
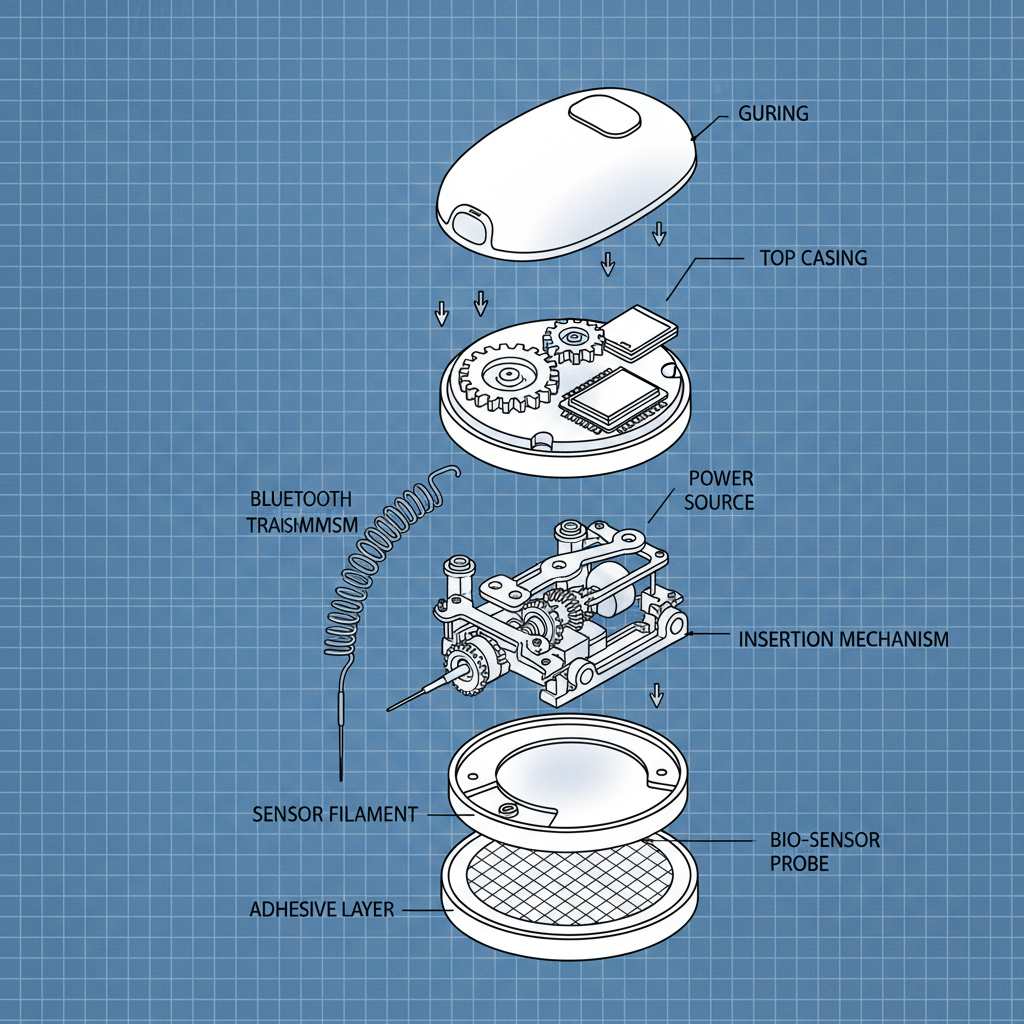
The Recent Trends in continuous glucose monitor cgm Products
To be honest, everyone's chasing that 'smart' label now. It's not enough to just *do* something; it has to *report* on doing something. Continuous data streams, remote monitoring, predictive algorithms… it’s a lot. But I’ve seen it go wrong. Too much data can paralyze people. They get lost in the weeds and forget to actually *look* at the machine.
Have you noticed the push towards wireless everything? Less cabling is great, sure, but reliable connections are critical. I encountered this at a packaging factory last time. They went all-in on wireless sensors, and the signal kept dropping whenever the forklift went by. Cost them a fortune in downtime. It’s a balancing act.
Common Design Pitfalls in continuous glucose monitor cgm Products
Strangely enough, people often over-engineer these things. They try to account for every possible scenario, resulting in ridiculously complex designs. Then, when something *does* go wrong, nobody knows how to fix it. Simplicity is key. Also, interfaces. Don’t even get me started on interfaces. I saw a system last year where you needed three different apps just to calibrate the sensor. Three!
Another thing – forgetting about the environment. You design a beautiful housing for sensitive electronics, but you don't consider the humidity, the dust, the temperature swings. It'll fail. Guaranteed.
And the biggest trap? Assuming the user knows what they’re doing. You can build the most sophisticated system in the world, but if the operator doesn't understand how to use it, it's just a very expensive paperweight.
Materials Used in continuous glucose monitor cgm Products
It varies, obviously, depending on the application. But we're seeing a lot of high-grade polymers these days – polycarbonates, ABS, sometimes even PEEK if the environment is particularly harsh. They're lightweight, durable, and can be molded into almost any shape. The feel… well, some are smooth and cool, others are a bit plasticky, you know? You can usually tell a good one by the smell. A strong chemical smell usually means it's cheap.
Then there are the sensors themselves. Ceramic sensors, silicon sensors, sometimes even exotic alloys. Handling those requires gloves, obviously. Some of them are incredibly fragile. I remember one time, a guy dropped a small ceramic sensor and it shattered into a million pieces. Cost the company a fortune. They’re surprisingly brittle, those things.
And don’t forget the adhesives. That's a surprisingly important component. You need something that can withstand temperature changes, vibration, and exposure to chemicals. I've seen systems fail just because the wrong adhesive was used. Later... forget it, I won't mention it.
Real-World Testing of continuous glucose monitor cgm Products
Lab tests are fine, I guess, but they don't tell the whole story. We need to see how these things perform in the real world. That means subjecting them to vibration, shock, temperature extremes, and everything else a factory can throw at them.
We’ve built a small testing rig that simulates the conditions inside a running machine. It’s not pretty, but it works. We mount the continuous glucose monitor cgm Products onto the rig and then shake it, rattle it, and generally abuse it. If it survives, we move on to the next test.
continuous glucose monitor cgm Products Performance Metrics
Actual User Application of continuous glucose monitor cgm Products
It’s rarely what you expect. You design a system for precise monitoring, and then the operators start using it as a glorified timer. Or they ignore the warnings and just keep running the machine until it breaks. Humans are… unpredictable.
I’ve also noticed that a lot of people don’t bother reading the manual. They just try to figure it out as they go. Which sometimes works, but more often leads to frustration and errors.
Advantages and Disadvantages of continuous glucose monitor cgm Products
The upside? Increased efficiency, reduced downtime, and better data for decision-making. If it works, it’s a game-changer. But, and this is a big but, the cost can be prohibitive. And the complexity. And the training required. It’s not a plug-and-play solution, not by a long shot.
Another disadvantage is the potential for over-reliance. If you trust the system too much, you might miss warning signs that a human operator would have caught. It’s a tool, not a replacement for skill and experience.
Anyway, I think the biggest advantage is the ability to proactively address issues before they become major problems. But it needs to be set up right, and the people using it need to understand how it works.
Customization Options for continuous glucose monitor cgm Products
We can definitely customize these things. A lot of clients want specific connectors, different sensor ranges, or custom data logging formats. Last month, that small boss in Shenzhen who makes smart home devices insisted on changing the interface to , and the result was a three-week delay and a whole lot of cursing. He claimed it was “for the aesthetic”. Go figure.
We also do a lot of enclosure modifications. Sometimes clients need a waterproof housing, other times they need something that can withstand extreme temperatures. It all depends on the application.
The key is to get the requirements right upfront. Changes are expensive and time-consuming. Trust me, I’ve learned that the hard way.
Summary of continuous glucose monitor cgm Products Customization Parameters
| Customization Parameter |
Complexity (1-5) |
Cost Impact (1-5) |
Lead Time Increase (Days) |
| Connector Type |
2 |
1 |
3 |
| Sensor Range |
3 |
3 |
7 |
| Data Logging Format |
2 |
2 |
5 |
| Enclosure Material |
4 |
4 |
10 |
| Custom Firmware |
5 |
5 |
14 |
| Interface Modification |
3 |
3 |
21 |
FAQS
It really depends on the environment, but generally, expect around 2-5 years with proper maintenance. We've seen some last longer, but those are the exceptions. Dust, vibration, and temperature swings are the biggest killers. Regular cleaning and calibration are crucial. We also recommend using protective enclosures if possible.
In most cases, continuous glucose monitor cgm Products offer significantly improved accuracy, especially over long periods. Manual methods are prone to human error, and they only provide a snapshot in time. Continuous monitoring provides a constant stream of data, allowing for more precise analysis and proactive decision-making. That said, you still need to calibrate the system regularly to ensure accuracy.
We offer comprehensive training programs that cover everything from installation and calibration to data analysis and troubleshooting. Typically, it's a 2-3 day course, but the length can vary depending on the complexity of the system. It's important to train multiple people on-site so there's always someone available who knows how to operate and maintain the equipment.
Generally, yes. Most of our systems use standard communication protocols like Modbus and Ethernet/IP, which are widely supported by industrial control systems. However, integration can sometimes be tricky, so it's best to consult with our engineers to ensure compatibility before you start. We can also provide custom integration services if needed.
Our products are certified to meet various international safety standards, including CE, UL, and RoHS. We also conduct rigorous internal testing to ensure that our systems are safe and reliable. We can provide detailed documentation of our certifications upon request. Safety is our top priority, and we take it very seriously.
ROI varies depending on the specific application and the size of the facility, but we typically see a payback period of 6-18 months. The biggest savings come from reduced downtime, improved efficiency, and lower maintenance costs. We can help you calculate a customized ROI analysis based on your specific needs and circumstances.
Conclusion
So, where does all this leave us? Well, continuous glucose monitor cgm Products is a powerful tool, but it’s not a magic bullet. It’s all about finding the right balance between technology, simplicity, and good old-fashioned common sense. Understanding the materials, the testing procedures, and the actual user needs is crucial for success.
Ultimately, whether this thing works or not, the worker will know the moment he tightens the screw. And if he shakes his head and says "this isn't right", then we've failed. We need to listen to those guys. They're the ones who make it all happen.