Bloodless glucose monitors Products represent a significant advancement in diabetes management, offering a less invasive and more comfortable alternative to traditional finger-prick methods. These innovative devices are gaining traction globally as the prevalence of diabetes continues to rise, impacting millions worldwide. The development and refinement of these technologies are crucial in improving patient compliance and overall quality of life for individuals living with diabetes. bloodless glucose monitors Products are reshaping how individuals monitor their glucose levels.
The global market for blood glucose monitoring systems is projected for substantial growth, driven by an aging population, increasing rates of obesity, and rising awareness regarding preventative healthcare. The demand for convenient and pain-free monitoring solutions is fueling innovation in the field of continuous glucose monitoring (CGM) and flash glucose monitoring (FGM). This growth is particularly prominent in developing countries where access to affordable and effective diabetes care is paramount.
Understanding the advancements in bloodless glucose monitors Products allows healthcare professionals and individuals alike to make informed decisions regarding diabetes management. These technologies are not just about convenience; they represent a fundamental shift towards personalized and proactive healthcare, ultimately aiming to minimize the long-term complications associated with diabetes and improve patient outcomes.


Bloodless glucose monitors Products encompass a range of technologies designed to measure glucose levels without the need for traditional finger-prick blood samples. These typically include continuous glucose monitoring (CGM) systems and flash glucose monitoring (FGM) devices. CGM systems utilize a small sensor inserted under the skin to continuously track glucose levels, transmitting data to a receiver or smartphone. bloodless glucose monitors Products are rapidly evolving.
FGM devices, on the other hand, require a sensor to be scanned with a reader to obtain a current glucose reading and trend information. Both technologies offer significant advantages over traditional methods, including reduced pain, improved data insights, and the ability to identify patterns in glucose levels. This data-driven approach empowers individuals with diabetes to make more informed decisions about their diet, exercise, and medication.
Diabetes is a global health crisis, affecting an estimated 537 million adults worldwide in 2021, according to the International Diabetes Federation. This number is projected to rise to 783 million by 2045. Effective glucose monitoring is paramount in managing diabetes and preventing long-term complications such as cardiovascular disease, kidney failure, and blindness. The increasing burden of diabetes underscores the urgent need for innovative and accessible monitoring solutions.
Bloodless glucose monitors Products address this need by offering a more patient-friendly and convenient approach to glucose monitoring. This can lead to improved adherence to monitoring regimens, which is critical for effective diabetes management. The accessibility of these technologies is particularly important in underserved populations and developing countries where access to healthcare may be limited. bloodless glucose monitors Products are essential for managing the rising numbers.
Furthermore, the data generated by these devices can be shared with healthcare providers, enabling more personalized and proactive care. This shift towards data-driven diabetes management has the potential to significantly improve patient outcomes and reduce the overall healthcare costs associated with diabetes.
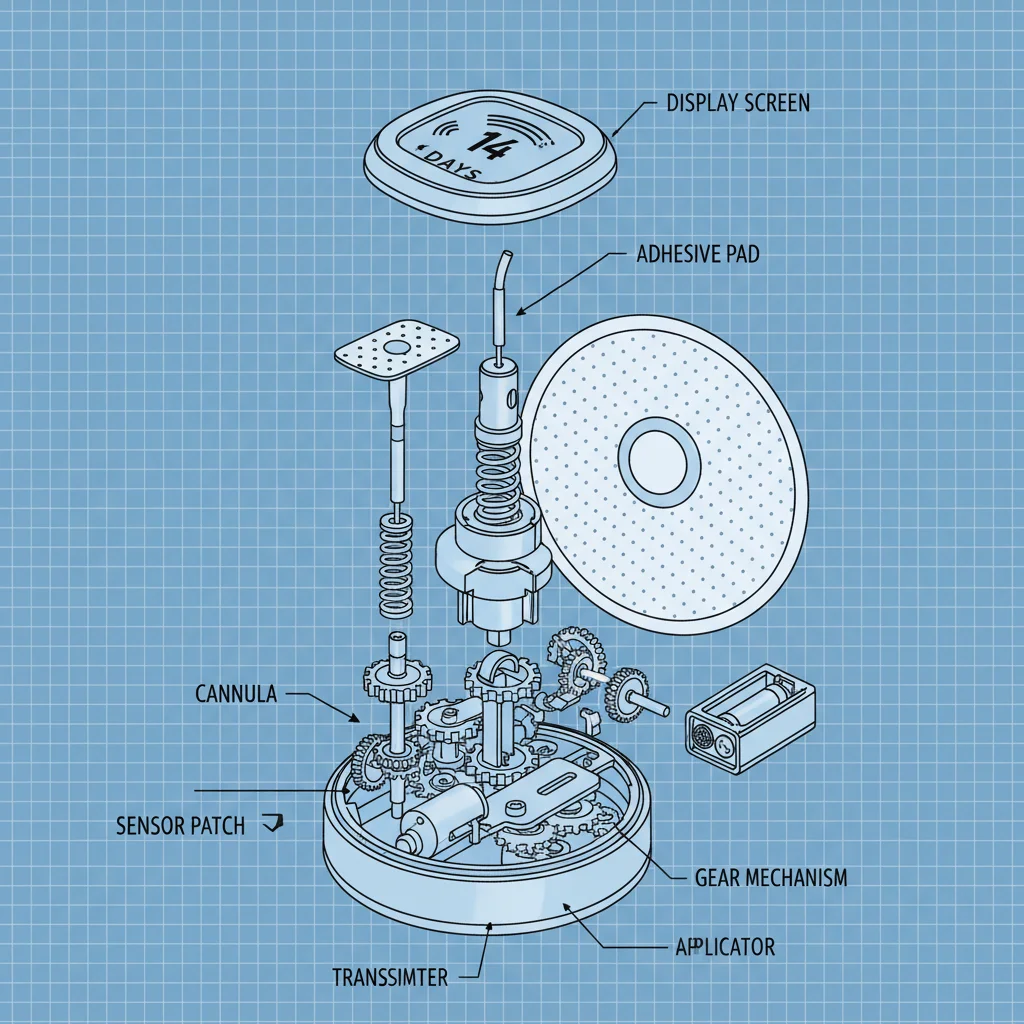
Bloodless glucose monitors Products can be broadly categorized into two main types: Continuous Glucose Monitoring (CGM) and Flash Glucose Monitoring (FGM). CGM systems use a small sensor inserted under the skin, typically in the abdomen or arm, to measure glucose levels in the interstitial fluid. These sensors transmit glucose readings to a receiver or smartphone app in real-time, providing a continuous stream of data.
FGM systems, like Abbott’s FreeStyle Libre, employ a sensor that is also inserted under the skin, but unlike CGM, the user needs to actively scan the sensor with a dedicated reader to obtain glucose readings. These readings are displayed on the reader and can be stored for trend analysis. FGM provides a snapshot of glucose levels and trends, offering valuable insights into glucose control.
Both CGM and FGM represent a significant evolution in glucose monitoring, offering individuals with diabetes a less invasive and more convenient way to manage their condition. They are integral to modern diabetes care, enabling better glycemic control, improved quality of life, and a proactive approach to preventing long-term complications.
Several key factors contribute to the effectiveness and usability of bloodless glucose monitors Products. Sensor accuracy is paramount, ensuring reliable glucose readings that patients can trust. Modern sensors employ advanced electrochemical technology to measure glucose levels with high precision.
Data transmission and connectivity are also crucial, allowing seamless communication between the sensor, receiver, and smartphone apps. Secure data storage and analysis features are essential for tracking trends, identifying patterns, and sharing information with healthcare professionals. bloodless glucose monitors Products offer various core components.
Bloodless glucose monitors Products are finding diverse applications in various healthcare settings and everyday life. In hospitals and clinics, they provide valuable data for managing patients with diabetes, optimizing insulin therapy, and preventing hypoglycemia. bloodless glucose monitors Products are widely used in clinical settings.
For individuals with Type 1 diabetes, CGM systems offer real-time insights into glucose fluctuations, enabling proactive adjustments to insulin doses and lifestyle choices. FGM devices are particularly useful for individuals with Type 2 diabetes who are not on insulin, providing a convenient way to monitor their glucose levels and make informed dietary and exercise decisions.
The advantages of bloodless glucose monitors Products extend beyond convenience and comfort. Improved glycemic control, reduced risk of hypoglycemia, and enhanced quality of life are key benefits. The continuous data stream provided by these devices empowers individuals to take a more active role in managing their diabetes.
Long-term, these technologies can significantly reduce the risk of diabetes-related complications, lowering healthcare costs and improving overall well-being. The ability to share data with healthcare providers facilitates more personalized and effective treatment plans, fostering a collaborative approach to diabetes care.
The field of bloodless glucose monitoring is rapidly evolving, with ongoing research focused on developing even less invasive and more accurate technologies. Non-invasive glucose monitoring, utilizing technologies like optical sensors and radiofrequency identification, holds immense promise for the future.
Integration with artificial intelligence (AI) and machine learning (ML) algorithms will enable predictive glucose monitoring, alerting users to potential fluctuations before they occur. Miniaturization of sensors and improved connectivity will further enhance the usability and convenience of these devices.
| Cost-Effectiveness | Ease of Use | Accuracy & Reliability | Data Connectivity & Analysis |
|---|---|---|---|
| Moderate (initial investment higher than traditional methods) | High (minimal intervention required) | Very High (accuracy continuously improving) | High (seamless data transfer to smartphones/cloud) |
| Long-Term Savings (reduced complications) | User-Friendly Interfaces | Regular Calibration Requirements | Data Security Concerns (privacy regulations) |
| Reimbursement Policies Impacting Affordability | Minimal Training Needed | Sensor Lifespan (replacement frequency) | Integration with EHR Systems (electronic health records) |
| Potential for Reduced Hospitalizations | Suitable for Various Age Groups | Environmental Factors Affecting Readings | Real-time Alerts and Notifications |
| Increased Patient Engagement & Adherence | Discreet Monitoring | Algorithm Updates for Improved Accuracy | Personalized Insights and Trend Analysis |
| Cost-Benefit Analysis Demonstrates Value | Reduced Pain and Anxiety | Sensor Insertion Site Sensitivity | Remote Monitoring Capabilities for Healthcare Providers |
CGM systems continuously monitor glucose levels and transmit data in real-time, while FGM requires scanning a sensor for readings. CGM provides a continuous stream of data, facilitating proactive management, whereas FGM offers on-demand insights. CGM generally has more frequent alerts and data points, but FGM is often more affordable and easier to initiate. Both bloodless glucose monitors Products offer substantial advantages over traditional methods.
Modern bloodless glucose monitors Products, particularly CGM systems, demonstrate high accuracy, comparable to or exceeding finger-prick testing in many cases. However, accuracy can be influenced by factors like sensor placement, calibration, and individual physiology. Regular calibration, as recommended by the manufacturer, is crucial for maintaining optimal accuracy. Ongoing advancements are continually improving the precision of these devices.
Generally, both CGM and FGM systems are suitable for individuals with Type 1 and Type 2 diabetes, but suitability depends on individual needs and treatment plans. CGM is often preferred by those on intensive insulin therapy, while FGM can be a valuable tool for individuals not on insulin. Consultation with a healthcare professional is essential to determine the most appropriate monitoring solution.
Sensor lifespan varies depending on the manufacturer and model, but typically ranges from 7 to 14 days. Once the sensor reaches the end of its lifespan, it needs to be replaced. Factors like sensor care and storage can impact performance and longevity. Adhering to the manufacturer’s guidelines regarding sensor replacement is critical for accurate and reliable glucose monitoring.
Insurance coverage for bloodless glucose monitors Products varies significantly depending on the insurance provider, plan type, and individual circumstances. Many insurance companies are now recognizing the value of these technologies and offering coverage, but pre-authorization and specific requirements may apply. It's crucial to verify coverage details with your insurance provider.
Potential limitations include skin irritation at the sensor insertion site, occasional inaccuracies, and the need for regular sensor replacements. Data overload can also be a challenge for some users. Proper training and ongoing support from healthcare professionals can help mitigate these challenges and ensure optimal device utilization.
Bloodless glucose monitors Products represent a transformative advancement in diabetes care, offering a less invasive, more convenient, and data-rich approach to glucose monitoring. These technologies empower individuals with diabetes to take control of their health, improve glycemic control, and reduce the risk of long-term complications. The continuous innovation in this field promises even more sophisticated and accessible monitoring solutions in the future.
As the prevalence of diabetes continues to rise, the adoption of bloodless glucose monitors Products will be crucial in improving patient outcomes and reducing the burden on healthcare systems. Further research and development, coupled with increased awareness and access, will be key to unlocking the full potential of these life-changing technologies. Visit our website to learn more about bloodless glucose monitors Products and find the right solution for your needs.



If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.
